To describe and analyse predictive and prognostic factors of overall survival (OS) in high-grade gliomas at our institution.
Material and methodAll patients diagnosed with grade III (GIII) or grade IV (GIV) gliomas (excluding oligodendrogliomas, oligoastrocytomas or infratentorial gliomas) were prospectively included from November 2010 to August 2014. All were treated with surgery followed by adjuvant radiochemotherapy. The Kaplan–Meier method was used for the statistical analysis, considering a p value <0.05 to be significant.
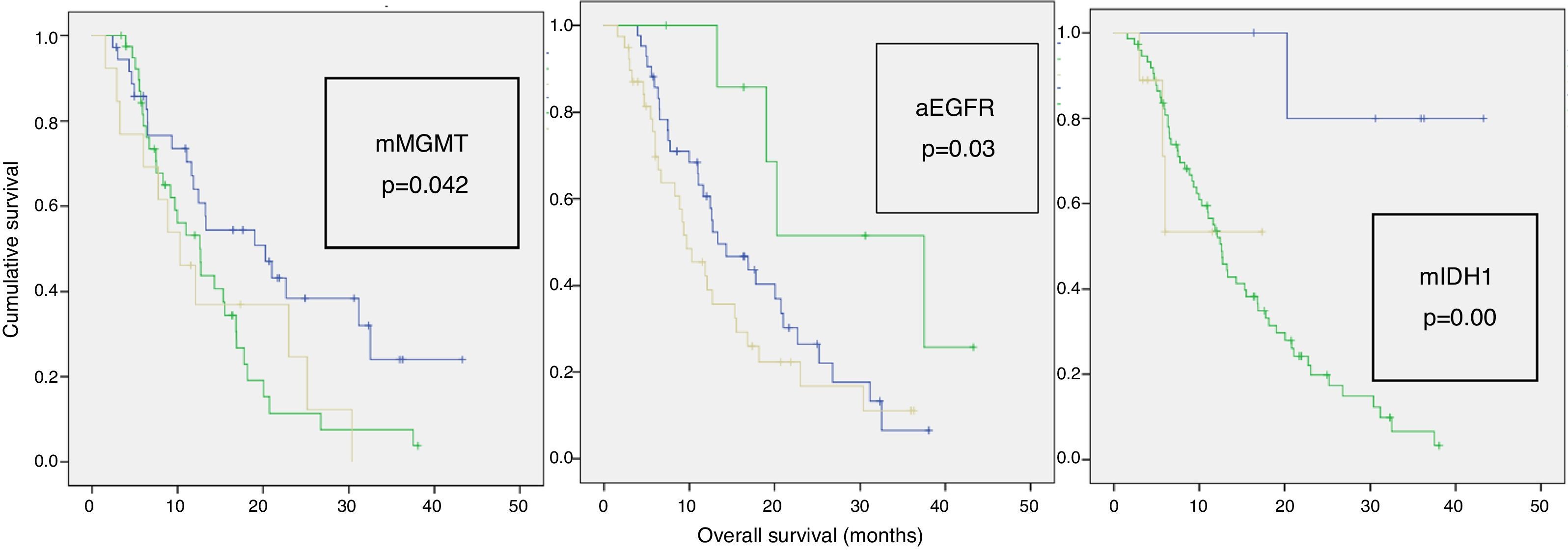
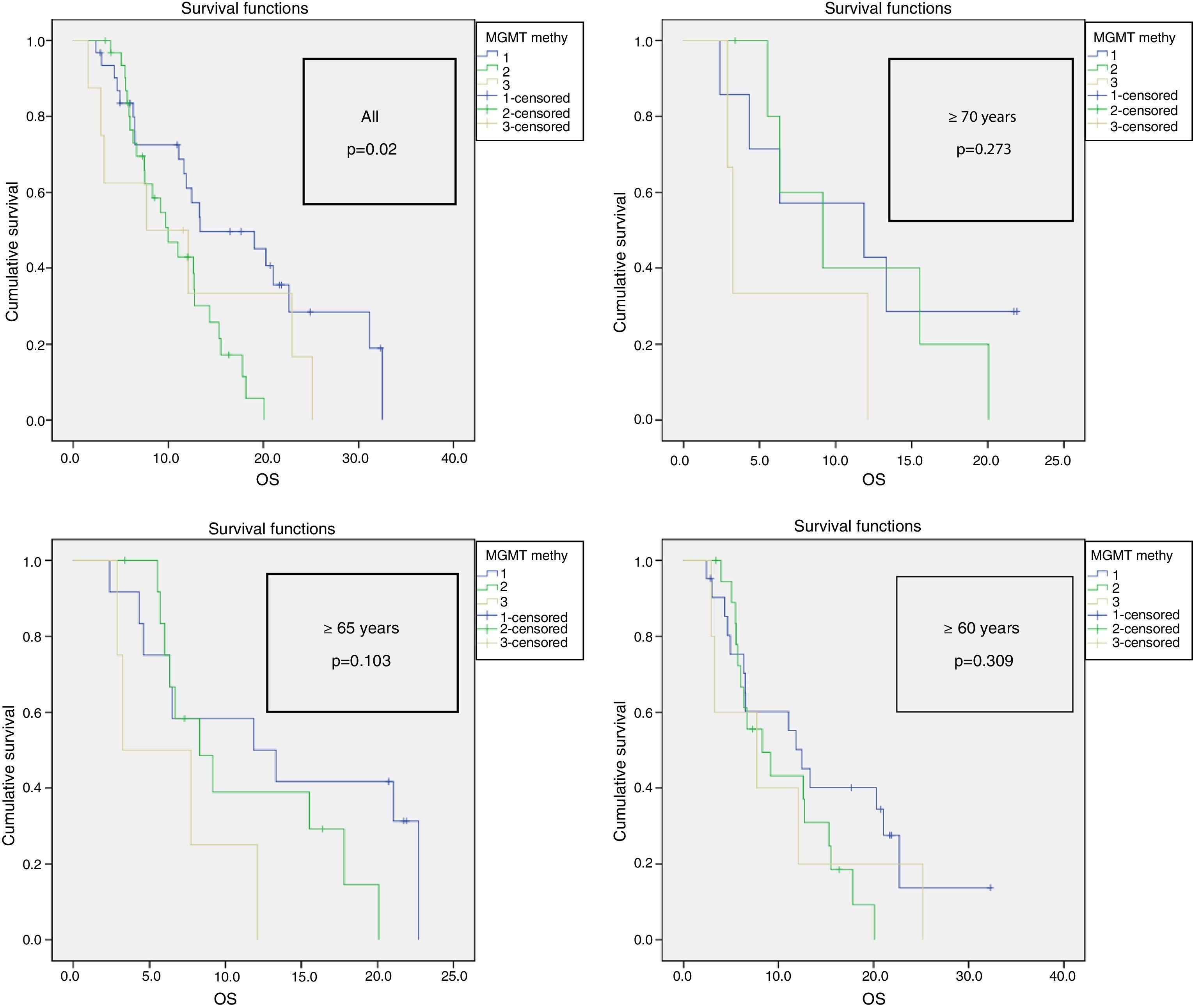
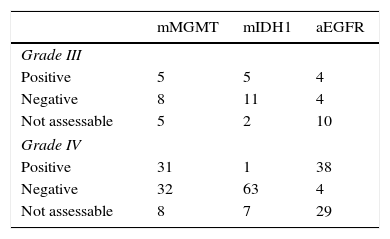
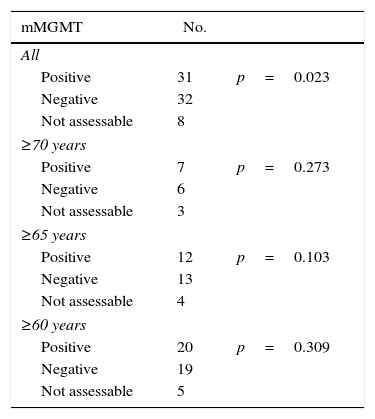
Results89 patients were studied (18 GIII and 71 GIV). The average age was 60 years and 55% were men. The mean Karnofsky score was 80%. The most common location was the frontal lobe (38%). A total of 65% were partial resections. Complete chemotherapy was administered to 74% and complete RT to 83% of patients. Mean OS was 26.8±8.3 months for GIII and 12.5±1 month for GIV. 72 had died by the end of this study. A total of 40% of patients had MGMT methylation, 7% IDH1 mutation and 47% EGFR amplification. Statistically significant variables for OS were: GIII (p=0.020), age <70 years (p=0.040), <65 years (p=0.013) and <60 years (p=0.003), Karnofsky≥70% (p=0.029), complete radiotherapy (p=0.000), complete resection (p=0.001), MGMT methylation (p=0.042), IDH1 mutation (p=0.007) and EGFR non-amplification (p=0.034). Additionally, GIII and GIV subgroups were independently analysed. In GIII, the only significant biomarker for OS was IDH1 mutation, while in GIV, MGMT methylation (p=0.023) was significant. Age ≥70 years old was a significant factor in the GIII-subgroup (p=0.040) but not for GIV (p=0.166).
ConclusionsData are in line with previous studies, with the exception of age, which does not appear to be significant in GIV.
Describir y analizar los factores predictivos y pronósticos en gliomas de alto grado respecto a supervivencia global (SG) en nuestro centro.
Material y métodoDesde noviembre drl 2010 hasta agosto del 2014 se incluyó prospectivamente a todos los pacientes diagnosticados de glioma grado III (GIII) o IV (GIV), excepto oligodendrogliomas, oligoastrocitomas e infratentoriales. Todos se trataron con cirugía y quimioradioterapia adyuvante. Análisis estadístico mediante el método de Kaplan-Meier considerando significativo un valor de p<0,05.
ResultadosSe estudió a 89 pacientes (18 GIII y 71 GIV). Edad media=60 años; el 55% hombres. El Karnofsky medio fue del 80%. La localización más frecuente (38%) fue frontal. El 65% realizó resección parcial. Se administró quimioterapia completa al 74% y radioterapia completa al 83%. La media de SG fue 26,8±8,3 meses en GIII y 12,5±1 mes en GIV; 72 habían fallecido al terminar el estudio. El 40% de los pacientes tenía MGMT-metilada, el 7% IDH1-mutado y el 47% EGFR-amplificado. Las variables estadísticamente significativas para SG fueron: GIII (p=0,020), edad < 70 (p=0,040), < 65 (p=0,013) y < 60 años (p=0,003), Karnofsky ≥ 70% (p=0,029), resección total (p=0,001), radioterapia completa (p=0,000), MGMT-metilada (p=0,042), IDH1-mutado (p=0,007) y EGFR-no amplificado (p=0,034). Los subgrupos GIII y GIV fueron analizados de manera independiente. En el GIII, el único biomarcador significativo para la SG fue IDH1-mutado y en el GIV la MGMT-metilada (p=0,023). La edad ≥ 70 años es significativa en el GIII (p=0,040) pero no para el GIV (p=0,166).
ConclusionesLos datos están en la línea de estudios anteriores, salvo la edad, que parece no decisiva en el GIV.
Article

If it is the first time you have accessed you can obtain your credentials by contacting Elsevier Spain in suscripciones@elsevier.com or by calling our Customer Service at902 88 87 40 if you are calling from Spain or at +34 932 418 800 (from 9 to 18h., GMT + 1) if you are calling outside of Spain.
If you already have your login data, please click here .
If you have forgotten your password you can you can recover it by clicking here and selecting the option ¿I have forgotten my password¿.